Introduction
Immune-mediated thrombotic thrombocytopenic purpura (iTTP) is a rare, life-threatening disease caused by a severe deficiency of a disintegrin and metalloproteinase with thrombospondin type 1 motif, member 13 (ADAMTS13). Caplacizumab is a von Willebrand factor-directed antibody fragment designed to treat iTTP in conjunction with therapeutic plasma exchange (TPE) and immunosuppressive therapy. Caplacizumab was approved in Europe in 2018, in the United States in 2019, and in Japan in 2022.
Methods
In the open-label, Phase 2/3 study (NCT04074187), Japanese patients ≥18 years old with a clinical diagnosis of iTTP and ≤1 prior TPE received caplacizumab in combination with TPE and immunosuppression for ≥30 days post-TPE and were followed for 4 weeks after the end of treatment. Patients with persistent ADAMTS13 deficiency (eg, <10%) were allowed treatment extension of up to 8 weeks. This post hoc analysis assessed outcomes including patient characteristics, time to platelet count recovery, time to normalization of all 3 organ damage markers (lactate dehydrogenase, cardiac troponin I, and serum creatinine), safety, time to recovery of ADAMTS13 activity, ADAMTS13 activity at end of treatment, and incidence of ADAMTS13 inhibitor elevation during treatment stratified by iTTP severity. Very severe iTTP at baseline was defined as follows: French severity score ≥3 or severe neurological involvement (eg, coma, seizures, focal deficit), or cardiac involvement (cardiac troponin level >2.5×ULN).
Results
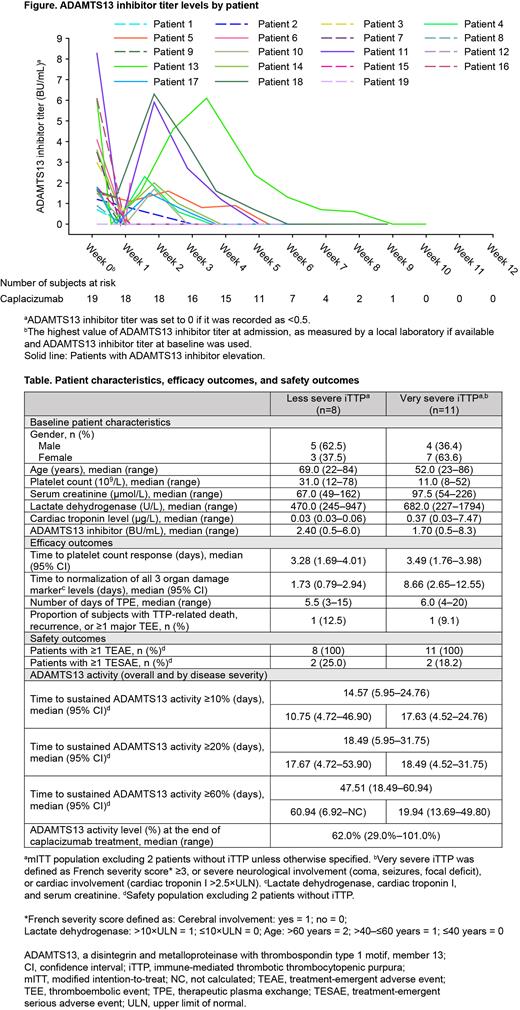
Overall, 21 patients were enrolled and treated with ≥1 dose of caplacizumab (modified intent-to-treat population [mITT] and safety population). Of these,19 patients had confirmed iTTP (less severe iTTP, n=8; very severe iTTP, n=11; Table).
Median (95% confidence interval [CI]) time to platelet count response and median (range) days of TPE was 3.28 (1.69-4.01) and 5.5 (3-15) days in patients with less severe iTTP and 3.49 (1.76-3.98) and 6.0 (4-20) days in patients with very severe iTTP, respectively. Median (95% CI) time to normalization of all 3 organ damage marker levels was 1.73 (0.79-2.94) days in patients with less severe iTTP and 8.66 (2.65-12.55) days in patients with very severe iTTP, which may reflect the fact that patients with very severe iTTP had worse organ damage at baseline. The number of patients experiencing recurrence, ≥1 major thromboembolic event, or TTP-related death in patients with less severe and very severe iTTP was 1 for each subgroup. Two patients in each disease severity subgroup had ≥1 treatment-emergent serious adverse event.
Of the 19 patients with confirmed iTTP, median (95% CI) time to recovery of ADAMTS13 activity to ≥10, ≥20, and ≥60% was 14.57 (5.95-24.76),18.49 (5.95-31.75), and 47.51 (18.49-60.94) days, respectively. Patients with ADAMTS13 inhibitor levels ≥2 BU/mL at baseline (n=7) took approximately twice as long to recover ADAMTS13 activity to ≥10, ≥20, and ≥60% versus patients with baseline inhibitor levels <2 BU/mL. Median (range) ADAMTS13 activity level at the end of caplacizumab treatment was 62.0% (29.0%-101.0%). ADAMTS13 inhibitor elevation occurred in 9 patients (Figure). Among them, 7 patients extended caplacizumab treatment. Only 1 patient with inhibitor re-increase experienced a recurrence 2 days after discontinuing caplacizumab due to an adverse event.
Conclusions
These results suggest that caplacizumab in conjunction with TPE and immunosuppression is effective for patients with iTTP regardless of their baseline severity and may reduce the risk of recurrence due to inhibitor re-increase.
Disclosures
Imada:Amgen K.K.: Honoraria; Sanofi K.K.: Honoraria; AbbVie GK.: Honoraria; Alexion Pharmaceuticals, Inc.: Honoraria; Nippon Kayaku Co., Ltd.: Honoraria; AstraZeneca K.K.: Honoraria; Towa Pharmaceutical Co., Ltd.: Honoraria; Janssen Pharmaceutical K.K.: Honoraria; Ono Pharmaceutical Co., Ltd.: Honoraria; Meiji Seika Pharma Co. Ltd.: Honoraria; Astellas Pharma Inc.: Honoraria; Otuka Pharmaceutical Co. Ltd.: Honoraria; Chugai Pharmaceutical Co., Ltd.: Honoraria; Nippon Shinyaku Co., Ltd.: Honoraria; Novartis Pharma K.K.: Honoraria; Takeda Pharmaceutical Co. Ltd.: Honoraria; Bristol-Myers Squibb K.K.: Honoraria; Kyowa Hakko Kirin Co., Ltd.: Honoraria. Miyakawa:Sanofi: Consultancy, Research Funding; Zenyaku Kogyo: Consultancy; argenx: Consultancy, Research Funding; Pfizer: Consultancy, Research Funding. Ueda:Sanofi: Honoraria, Speakers Bureau; AbbVie: Honoraria; Novo Nordisk: Honoraria. Hashimoto:Sanofi K.K.: Current Employment. Nishimi:Sanofi: Current Employment. Tahara:Sanofi K.K.: Current Employment. Matsumoto:Asahikasei Pharma: Research Funding, Speakers Bureau; Alexion Pharma: Consultancy, Research Funding, Speakers Bureau; Chugai Pharmaceutical: Research Funding; Takeda: Consultancy, Speakers Bureau; Sanofi: Consultancy, Research Funding, Speakers Bureau.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal